
FDA Advisors' Recommendation on Renal Denervation Hinges on
Price: $ 26.00
5(90)
Device-based hypertension treatments have only modest BP reductions

RQM+ on LinkedIn: #medtech #labdevelopedtests #ldtregulations

Therapeutic Strategies to Target the Androgen Receptor

Mechanisms and pharmacotherapy of hypertension associated with type 2 diabetes - ScienceDirect
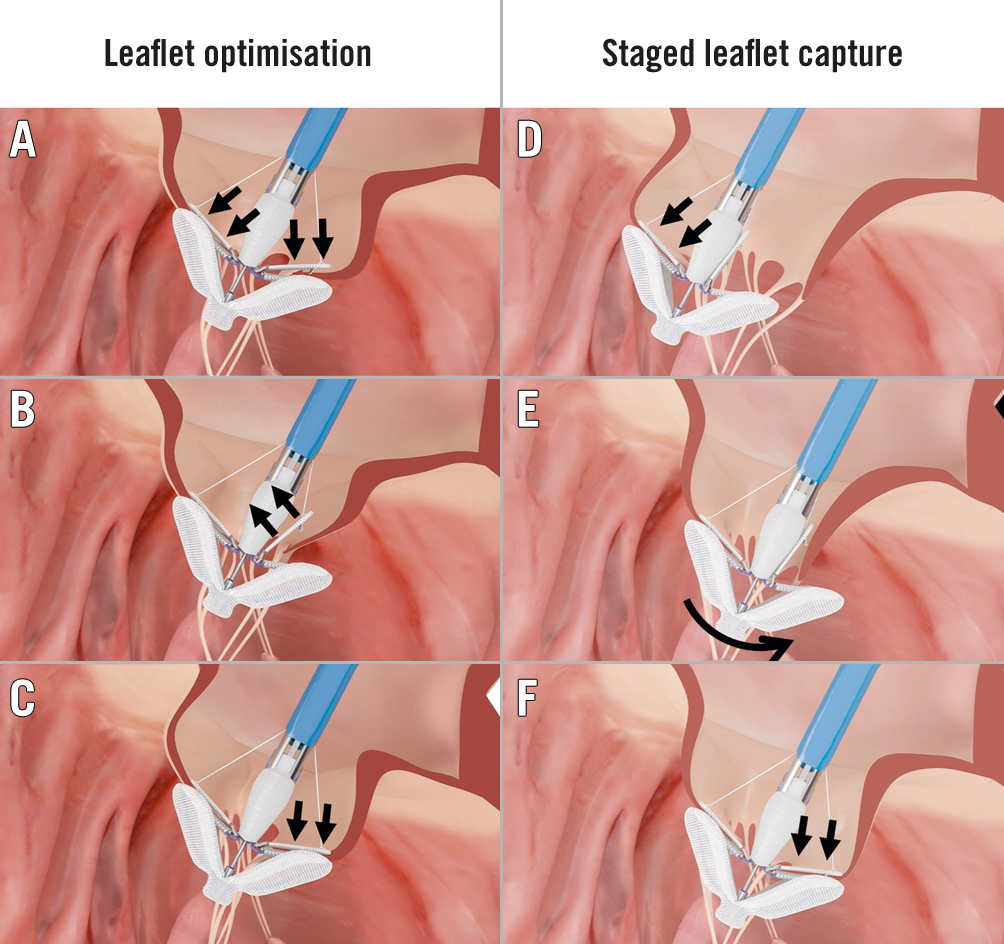
Mitral valve transcatheter edge-to-edge repair - EuroIntervention

Interventional News Issue 75 ROW by BIBA Publishing - Issuu
FDA Panel Split on Efficacy of Spyral Renal Denervation
FDA panel split on risk and benefit of Symplicity Spyral renal denervation system

ECR 13.1 by Radcliffe Cardiology - Issuu
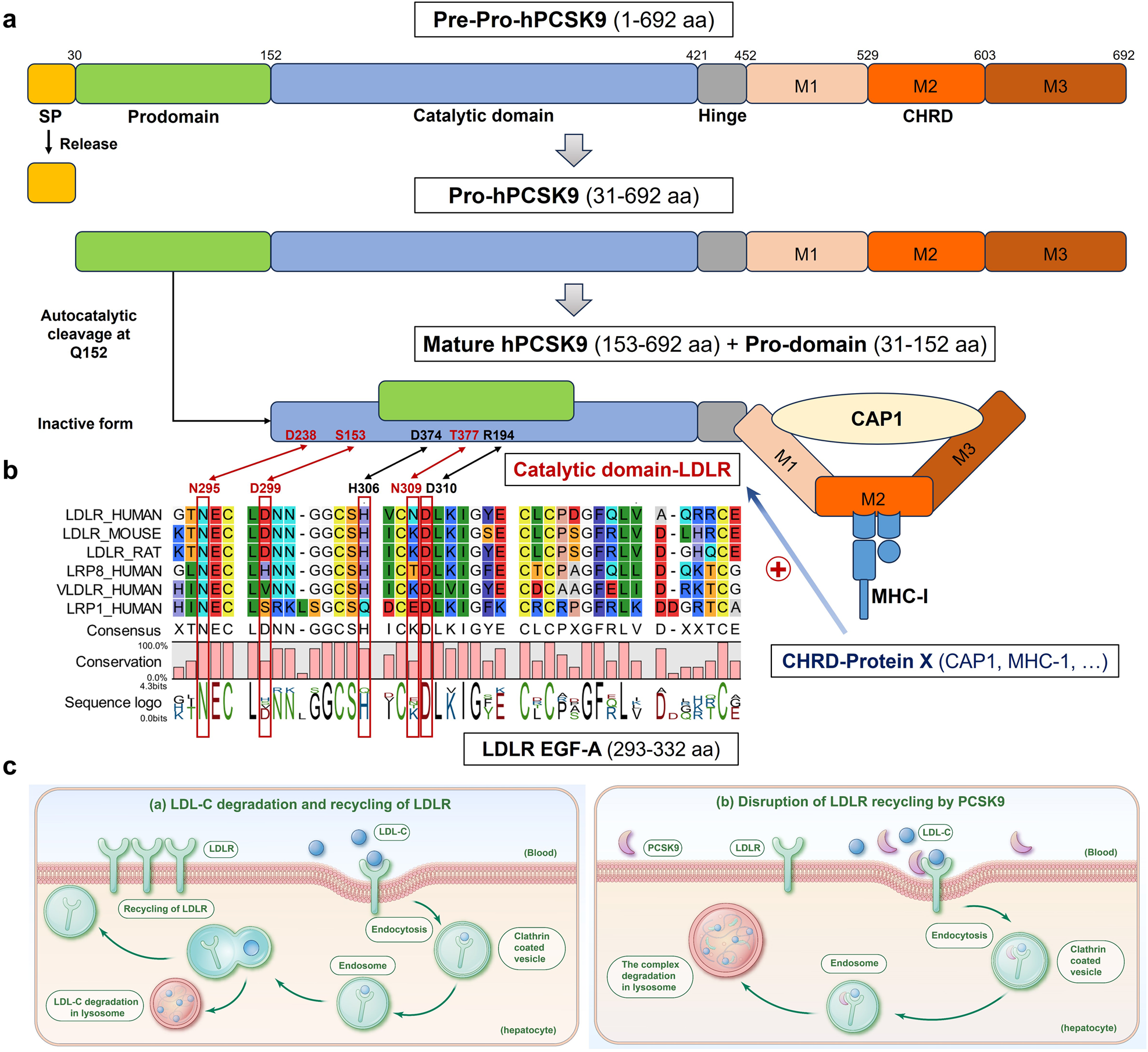
Targeting proprotein convertase subtilisin/kexin type 9 (PCSK9): from bench to bedside
First FDA Approval of Renal Denervation System to Treat Hypertension Announced by Recor Medical and Otsuka Medical Devices

FDA panel recommends ultrasound renal denervation device for uncontrolled hypertension
FDA approval to enrol first US renal denervation study for patients with moderate uncontrolled hypertension - Interventional News




