
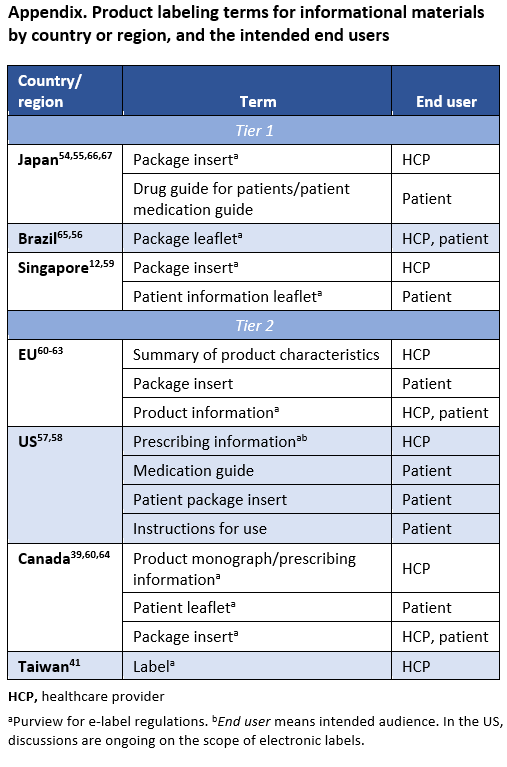
HSA Draft Guidance on Clinical Evaluation: Definitions
Price: $ 46.50
5(138)
The document describes in detail the aspects to be taken into account when preparing a site master file.
E-labeling and digital transformation in healthcare

HSA's Medical Device Evaluation Routes Freyr - Global Regulatory Solutions and Services Company

Modulation of Triple Artemisinin-Based Combination Therapy Pharmacodynamics by Plasmodium falciparum Genotype

HSA Guidance on Essential Principles: Overview

Ultimate Guide to Clinical Evaluation of a Medical Device in the EU

New FDA guidance offers ethical roadmap for including children in clinical trials

New HSA Guidance on UDI: Overview

HSA Guidance on Change Notification: Overview
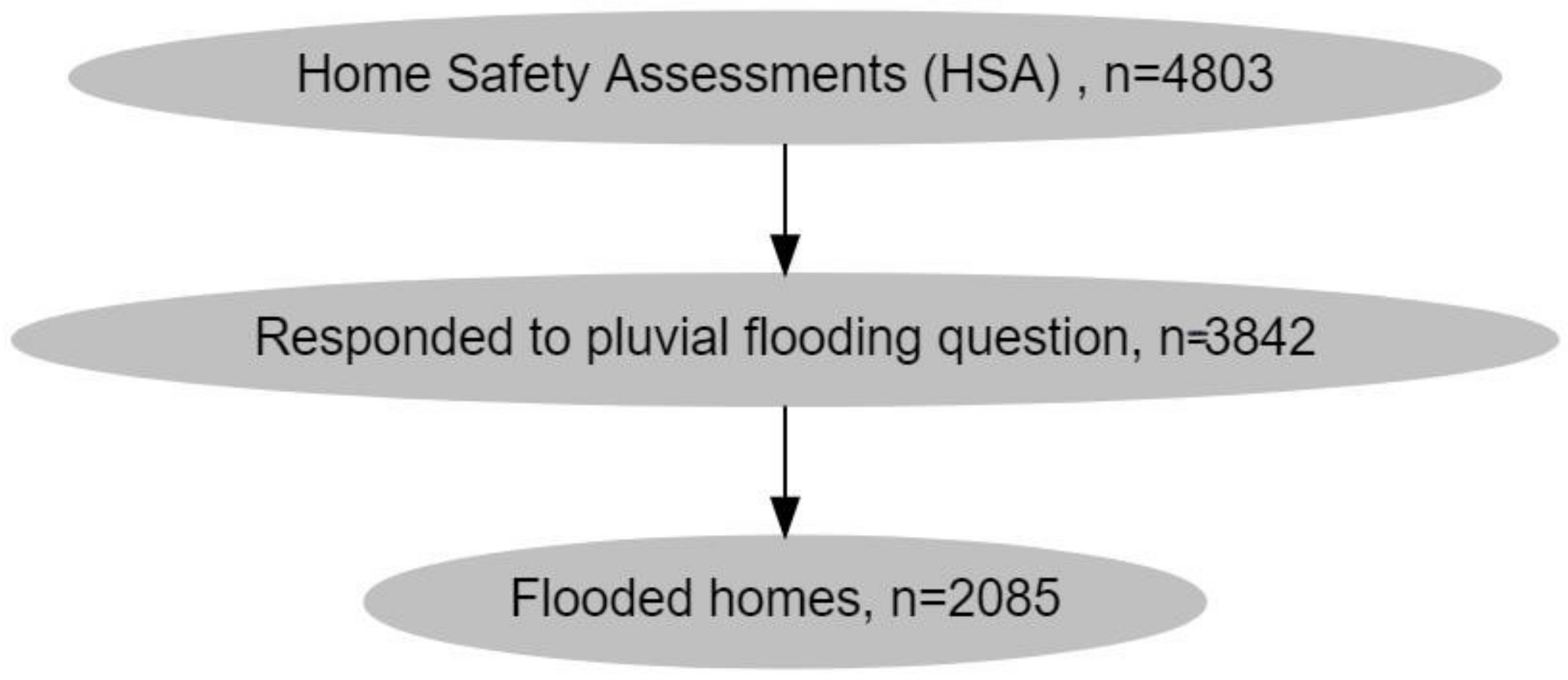
IJERPH, Free Full-Text
HSA Draft Guidance on Clinical Evaluation: Definitions

HSA Guidance on Clinical Evaluation: Overview

Singapore: Feedback Revised Draft of GN-20: Clinical Evaluation

HSA Guidance on Medical Device Registration: Basics

MicroRNA hsa-miR-320a-3p and Its Targeted mRNA FKBP5 Were Differentially Expressed in Patients with HIV/TB Co-Infection

Pooled depot clearance data (plotted as mean natural logarithm [ln] of


:max_bytes(150000):strip_icc():focal(599x0:601x2)/dr-squatch-pine-tar-soap-tout-29b3fe14756445bf8548c9a42136a24f.jpg)

