
Does Fitbit's WiFi-enabled Aria scale need FDA clearance?
Price: $ 42.99
4.9(735)
In January 2012 Fitbit expanded beyond wearable activity tracking devices when it launched its Aria WiFi Smart Scale at the Consumer Electronics Show (CES). While it offers much of the same functionality -- if not identical features -- to many of its peers, it is now one of the only connected weight scales that does not have FDA 510(k) clearance as a medical device. Perhaps the best known WiFi-enabled weight scale is Withings', which for a number of years sold through various retail channels, including Apple Stores, without a 510(k) clearance.

So What Does Google Want With Fitbit?
14 Best Fitness Trackers of 2024 - Reviewed
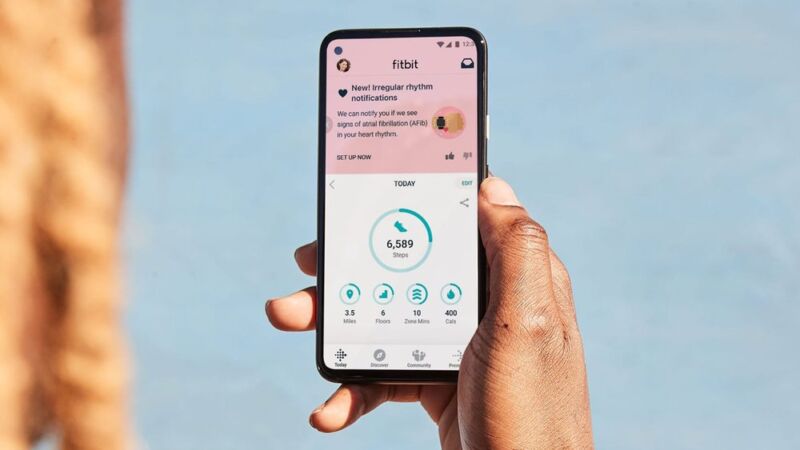
Fitbit gains FDA clearance for new atrial fibrillation-detection feature

Fitbit Aria Wi-Fi Smart Scale, Black : Electronics

When Your Fitbit Goes From Activity Tracker to Personal Medical Device

Tracking Your Health? Premium Fitbit Sense Smartwatch Has What You Need

Fitbit Aria Air smart scales review - Saga Exceptional

Fitbit and Apple know their smartwatches aren't medical devices. But do you? - CNET

Fitbit Receives Regulatory Clearance in Both the United States and Europe for ECG App to Identify Atrial Fibrillation (AFib)

Fitbit Aria Air smart scales review - Saga Exceptional

Irregular Rhythm

Fitbit Aria Wi-Fi Smart Scale review

Fitbit Aria Wi-Fi Smart Scale, Black : Electronics




