
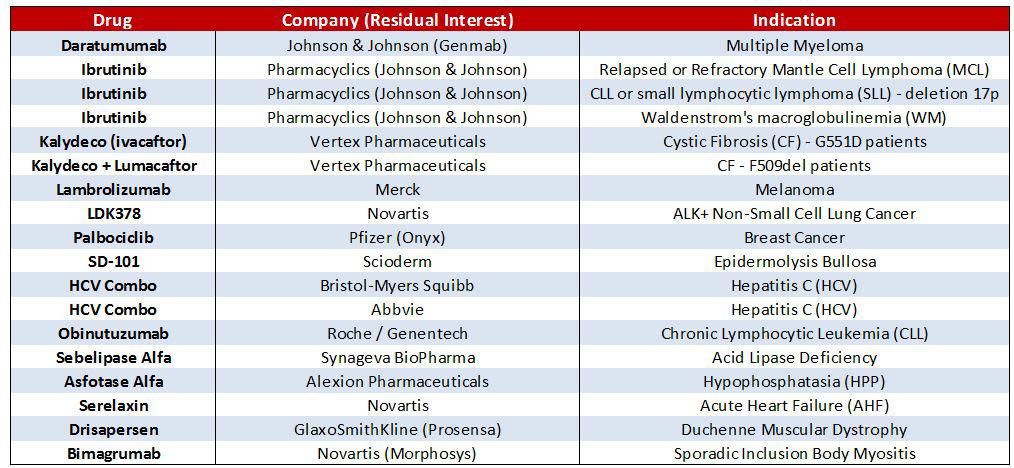
FDA Grants Fast Track to Palisade Bio's Drug Candidate
Price: $ 99.99
4.9(260)
FDA Grants Fast Track to Palisade Bio’s Drug Candidate
What Does The FDA's Breakthrough Designation Mean For Investors?

SEC Filing Vistagen Therapeutics, Inc

Medical Publication by Paul Streeto - Issuu

FDA news: Fast track for glioblastoma drug, breakthrough designation for leukemia therapy

10-K

Fast-Track Drug Approval, Designed for Emergencies, Is Now Routine - WSJ

BioVie Investor Deck, March 2021

Prevail Therapeutics' Parkinson's drug gets US FDA fast track designation

In Vitro and In Vivo Toxicity and Biodistribution of Paclitaxel-Loaded Cubosomes as a Drug Delivery Nanocarrier: A Case Study Using an A431 Skin Cancer Xenograft Model
vtgn20220331_10k.htm
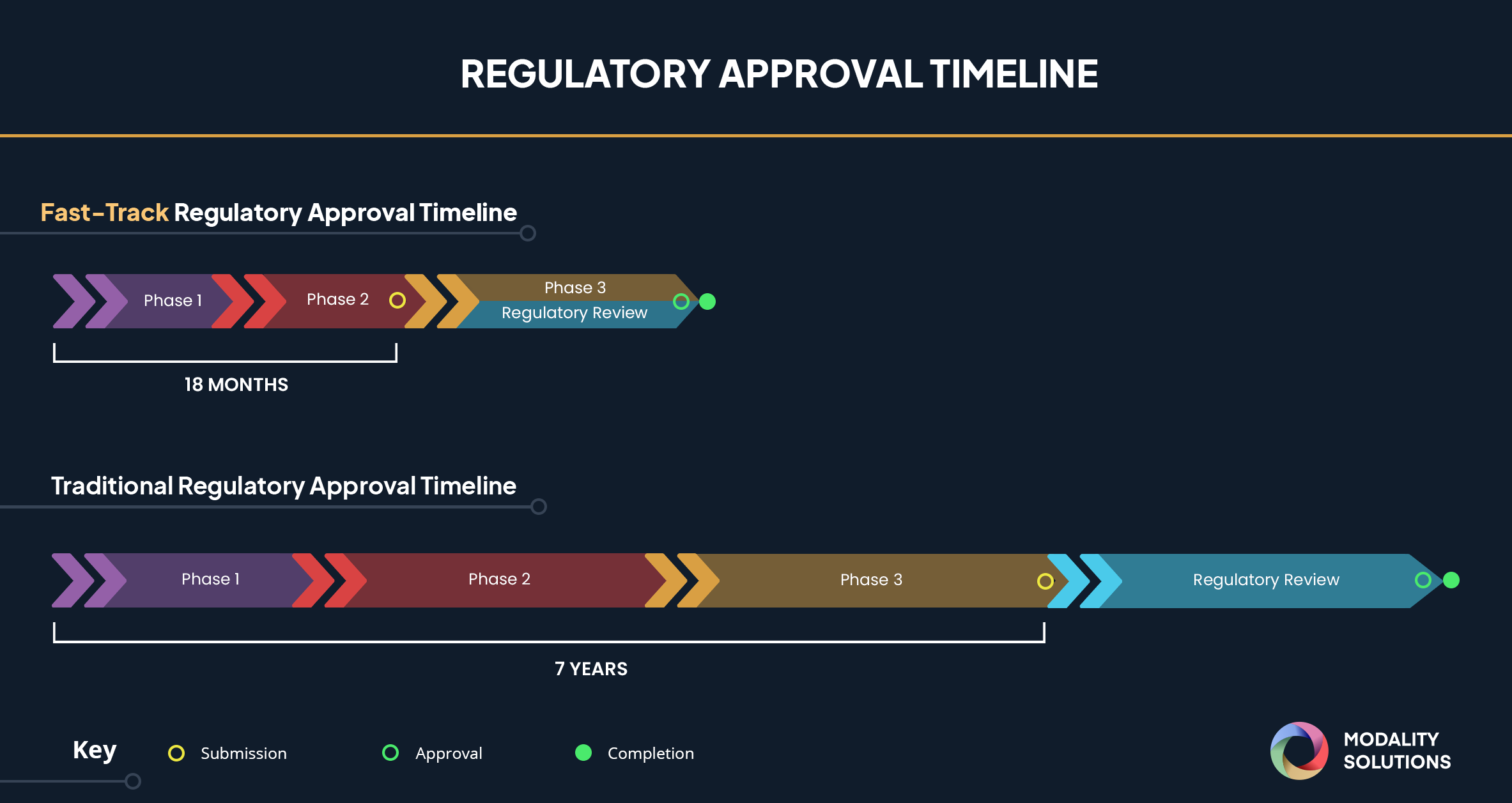
FDA's Fast Track Approval Coronavirus Treatment Acceleration Program
Press Releases - Palisade Bio

Seneca Biopharma Inc - 10K - Annual Report - March 17, 2022

BioVie Investor Deck, March 2021

Venture-Backed — PortfolioList




