
Side-chain hydrophobicity scale derived from transmembrane protein folding into lipid bilayers
Price: $ 33.99
4.8(458)

Lipid bilayer induces contraction of the denatured state ensemble of a helical-bundle membrane protein

Systematic Design and Validation of Ion Channel Stabilization of Amphipathic α-Helical Peptides Incorporating Tryptophan Residues

Energetics of side-chain partitioning of β-signal residues in unassisted folding of a transmembrane β-barrel protein - ScienceDirect
Chapter 6 - Cell and Organelle Membrane Structure” in “Fundamentals of Cell Biology”

Hydrophobicity scales - Wikipedia

Structure

Role of the lipid bilayer in outer membrane protein folding in Gram-negative bacteria - ScienceDirect

The energetics of transmembrane helix insertion and the consequences

Figure 5 from Curvature Forces in Membrane Lipid − Protein Interactions

Curvature Forces in Membrane Lipid–Protein Interactions
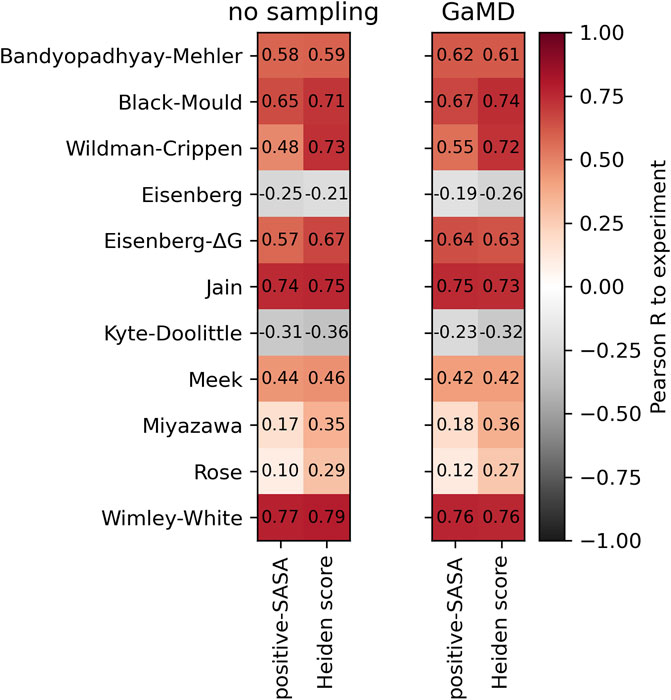
Frontiers Comparison of hydrophobicity scales for predicting biophysical properties of antibodies




